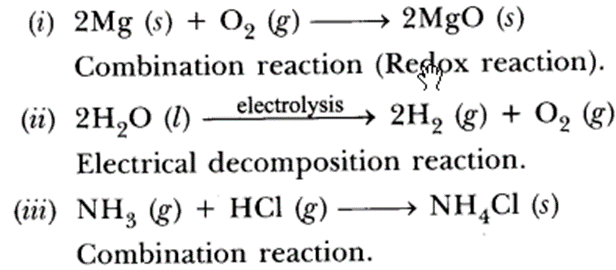Question 1.
Identify ‘x’, ‘y’ and ‘z’ in the following reaction:
(a) x = gas; y = reaction condition; z = gas
(b) x = solid; y = liquid; z = gas
(c) x = number of moles of KClO3; y = reaction condition; z = number of molecules of oxygen
(d) x = physical state of KClO3 and KCl;
y = reaction condition, z = physical state of O2. (2020)
Answer:

Question 2.
Assertion (A) : Following is a balanced chemical equation for the action of steam on iron : 3Fe + 4H2O → Fe3O4 + 4H2
Reason (R): The law of conservation of mass holds good for a chemical equation.
(a) Both (A) and (R) are true and reason (R) is the correct explanation of the assertion (A)
(b) Both (A) and (R) are true, but reason (R) is not the correct explanation of the assertion (A).
(c) (A) is true, but (R) is false.
(d) (A) is false, but (R) is true. (2020)
Answer:
A balanced chemical equation must obey the law of conservation of mass.
Question 3.
(a) State the law that is followed by balancing a chemical equation.
(b) Balance the following chemical equation: Na + H3O → NaOH + H2 (Board Term I, 2013)
Answer:
(a) Law of conservation of mass is followed for balancing a chemical equation which states that mass can neither be created nor destroyed in a chemical reaction. That is, the total mass of the elements present in the products of a chemical reaction has to be equal to the total mass of the elements present in the reactants in a balanced equation.
(b) 2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g)
Question 4.
Explain the significance of photosynthesis.
Write the balanced chemical equation involved in the process. (Board Term I, 2017)
Answer:
Photosynthesis means synthesis with the help of light. It is the process that gives life to all living beings.
Photosynthesis is a process by which plants utilize carbon dioxide and water in the presence of sunlight to produce glucose and oxygen.
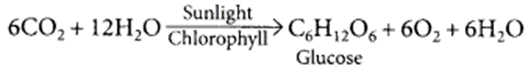
Question 5.
Write balanced chemical equations for the following chemical reactions:
(a) Hydrogen + Chlorine → Hydrogen chloride
(b) Lead + Copper chloride → Lead chloride + Copper
(c) Zinc oxide + Carbon → Zinc + Carbon monoxide (Board Term I, 2014)
Answer:
(a) H2(g) + Cl2(g) → 2HCl(g)
(b) Pb(s) + CuCl2(aq) → PbCl2(aq)+ Cu(s)
(c) ZnO(s) + C(s) → Zn(s) + CO(g)
Question 6.
Calcium oxide reacts vigorously with water to produce slaked lime.
CaO(s) + H2O(l) → Ca (OH)2(aq)
This reaction can be classified as
(A) Combination reaction
(B) Exothermic reaction
(C) Endothermic reaction
(D) Oxidation reaction
Which of the following is a correct option? (2020)
(a) (A) and (C)
(b) (C) and (D)
(c) (A), (C) and (D)
(d) (A) and (B)
Answer:
(d) The reaction between CaO and H2O to form Ca (OH)2 is an exothermic combination reaction.
Question 7.
When hydrogen sulphide gas is passed through a blue solution of copper sulphate, a black precipitate of copper sulphide is obtained and the sulphuric acid so formed remains in the solution. The reaction is an example of a (2020)
(a) combination reaction
(b) displacement reaction
(c) decomposition reaction
(d) double displacement reaction.
Answer:
(d) CuSO4 + H2S → CuS + H2SO4
It is a double displacement reaction as in this reaction CuSO4 and H2S reacting by exchange of Cu2+ and H+ ions to from two new compounds i.e., CuS and H2SO4.
Question 8.
In a double displacement reaction such as the reaction between sodium sulphate solution and barium chloride solution :
(A) exchange of atoms takes place
(B) exchange of ions takes place
(C) a precipitate is produced
(D) an insoluble salt is produced
The correct option is (2020)
(a) (B) and (D)
(b) (A) and (C)
(c) only (B)
(d) (B), (C) and (D)
Answer:
(d) In this reaction exchange of Na+ and Ba2+ ions takes place forming BaSO4 which is a white precipitate i.e., an insoluble salt.
Na2SO4 + BaCl2 → BaSO4 ↓+ 2NaCl
Question 9.
In which of the following, the identity of initial substance remains unchanged? (2020)
(a) Curdling of milk
(b) Formation of crystals by process of crystallisation
(c) Fermentation of grapes
(d) Digestion of food
Answer:
(b): Formation of crystals is a physical change rest others are chemical change.
Question 10.
Study the following equation of a chemical reaction: (Board Term 1, 2015)
H2 + Cl2 → 2HCl
(i) Identify the type of reaction.
(ii) Write a balanced chemical equation of another example of this type of reaction.
Answer:
(i) Combination reaction.
(ii) Another example of combination reaction is

Question 11.
State the type of chemical reactions, represented by the following equations: (Board Term I, 2014)
(a) A + BC → AC + B
(b) A + B → C
(c) PQ + RS → PS + RQ
(d) A2O3 + 2B → B2O3 + 2A
Answer:
(a) Displacement reaction.
(b) Combination reaction.
(c) Double displacement reaction.
(d) Displacement reaction or redox reaction.
Question 12.
1 g of copper powder was taken in a China dish and heated. What change takes place on heating? When hydrogen gas is passed over this heated substance, a visible change is seen in it. Give the chemical equations of reactions, the name and the colour of the products formed in each case. (2020)
Answer:
When copper powder is heated in a China dish, the reddish-brown surface of copper powder becomes coated with a black substance which is copper oxide.
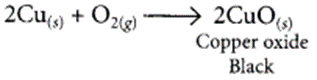
When hydrogen gas is passed over CuO, the black coating on the surface turned reddish brown due to the formation of Cu.
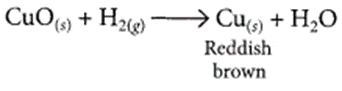
Question 13.
A compound ‘A’ is used in the manufacture of cement. When dissolved in water, it evolves a large amount of heat and forms compound ‘B’.
(i) Identify A and B.
(ii) Write chemical equation for the reaction of A with water.
(iii) List two types of reaction in which this reaction may be classified. (2020)
Answer:
(i) A is calcium oxide, CaO which is used in the manufacturing of cement.
B is calcium hydroxide Ca (OH)3.

(iii) The given reaction is a combination reaction.
Example : NH3(g)(g) + HCl(g) → NH4Cl(s)
2NO(g) + 02(g) → 2NO2(g)
Question 14.
Mention with reason the colour changes observe when:
(i) silver chloride is exposed to sunlight.
(ii) copper powder is strongly heated in the presence of oxygen.
(iii) a piece of zinc is dropped in copper sulphate solution. (2020)
Answer:
(i) When white silver chloride is left exposed to sunlight, its colour changes to grey as it decomposes to silver in the presence of sunlight.

This type of reaction is called photodecomposition reaction.
(ii) When copper powder is strongly heated in presence of oxygen, the reddish-brown surface of copper powder becomes coated with a black substance which is copper oxide.
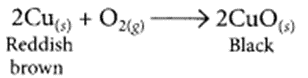
(iii) The blue color of copper sulphate fades gradually when a piece of zinc is dropped in copper sulphate solution, due to the formation of colorless zinc sulphate solution and deposition of reddish-brown copper metal on the zinc piece.
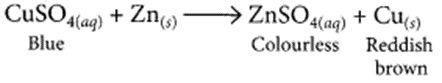
Question 15.
Lead nitrate solution is added to a test tube containing potassium iodide solution.
(a) Write the name and colour of the compound precipitated.
(b) Write the balanced chemical equation for the reaction involved.
(c) Name the type of this reaction justifying your answer. (2020)
Answer:
(a) When lead nitrate is added to potassium iodide then yellow precipitate of lead iodide is formed along with potassium nitrate.
(b) Balanced chemical reaction is as follows:
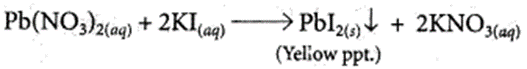
(c) This type of reaction is called precipitation reaction in which one of the products formed is an insoluble substance or this is also called double displacement reaction.
Question 16.
2 g of silver chloride is taken in a China dish and the China dish is placed in sunlight for some time. What will be your observation in this case? Write the chemical reaction involved in the form of a balanced chemical equation. Identify the type of chemical reaction. (Delhi 2019)
Answer:
Refer to answer 14(i).
Question 17.
Identify the type of reactions taking place in each of the following cases and write the balanced chemical equation for the reactions.
(a) Zinc reacts with silver nitrate to produce zinc nitrate and silver.
(b) Potassium iodide reacts with lead nitrate to produce potassium nitrate and lead iodide. (Delhi 2019)
Answer:
(a) It is a displacement reaction.
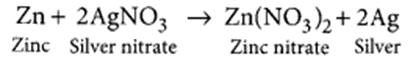
(b) Refer to answer 15.
Question 18.
2 g of ferrous sulphate crystals is heated in a dry boiling tube. (Al 2019, Board Term 1, 2017, 2016)
(a) List any two observations.
(b) Name the type of chemical reaction taking place.
(c) Write balanced chemical equation for the reaction and name the products formed.
Answer:
(a) Ferrous sulphate crystals (FeSO4.7H2O) lose water when heated and the colour of the crystal’s changes. It then decomposes to ferric oxide (Fe2O3), sulphur dioxide (SO2) and sulphur trioxide (SO3) with a smell of burning sulphur.
(b) This is a thermal decomposition reaction.
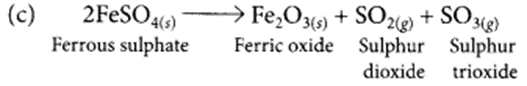
Question 19.
You might have noted that when copper powder is heated in a China dish, the reddish brown surface of copper powder becomes coated with a black substance. (AI 2019)
(a) Why has this black substance formed?
(b) What is the black substance?
(c) Write the chemical equation of the reaction that takes place.
(d) How can the black coating on the surface be turned reddish brown?
Answer:
(a) The black substance is formed because copper combines with oxygen.
(b) The black substance is copper oxide (CuO).
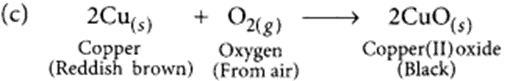
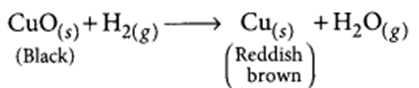
(d) The black coating on the surface can be turned reddish brown by passing hydrogen gas over the hot copper oxide.
Question 20.
Decomposition reactions require energy either in the form of heat or light or electricity for breaking down the reactants. Write one equation each for decomposition reactions where energy is supplied in the form of heat, light and electricity. (2018)
Answer:
Decomposition reaction involving absorption of heat:

Decomposition reaction involving absorption of light:
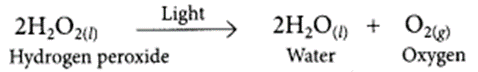
Decomposition reaction involving absorption of electrical energy:

Question 21.
Take 3 g of barium hydroxide in a test tube, now add about 2 g of ammonium chloride and mix the contents with the help of a glass rod. Now touch the test tube from outside.
(i) What do you feel on touching the test tube?
(ii) State the inference about the type of reaction occurred.
(iii) Write the balanced chemical equation of the reaction involved. (Board Term I, 2017)
Answer:
(i) The test tube bottom becomes cooler when barium hydroxide is added to ammonium chloride.
(ii) It is an endothermic reaction.
(iii) Ba(OH)2 + 2NH4Cl → BaCl2 + 2NH4OH
Question 22.
(a) When silver nitrate solution is mixed with a solution of potassium chloride, a white insoluble substance is formed. Write the chemical reaction involved and also mention the type of the chemical reaction.
(b) Ferrous sulphate when heated, decomposes with the evolution of a gas having a characteristic odour of burning sulphur. Write the chemical reaction involved and identify the type of reaction. (Board Term I, 2016)
Answer:
(a)

It is a double displacement reaction.
(b) Refer to answer 18(b) and (c).
Question 23.
Name the type of chemical reaction represented by the following equation: (Board Term I, 2016)
(i) CaO + H2O → Ca(OH)2
(ii) 3BaCl2 + Al2(SO4)3 → 2AlCl3 + 3BaSO4
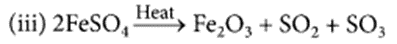
Answer:
(i) Combination reaction.
(ii) Precipitation reaction or double displacement reaction.
(iii) Thermal decomposition reaction.
Question 24.
What is a reduction reaction?
Identify the substances that are oxidised and the substances that are reduced in the following reactions. (Board Term I, 2015)
(a) Fe2O3 + 2Al → Al2O3 + 2Fe
(b) 2PbO + C → 2Pb + CO2
Answer:
Those reactions in which addition of hydrogen to a substance or removal of oxygen from a substance take place are called reduction reactions.
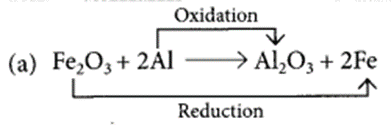
Fe2O3 is getting reduced to Fe and Al is getting oxidised to Al2O3.
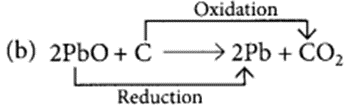
PbO is reduced to Pb and C is oxidised to CO2.
Question 25.
(a) Can a displacement reaction be a redox reaction? Explain with the help of an example.
(b) Write the type of chemical reaction in the following:
(i) Reaction between an acid and a base
(ii) Rusting of iron. (Board Term I, 2017)
Answer:
(a) Consider the following displacement reaction:
Zn(s)+ CuSO4(aq) → ZnSO4(aq) + Cu(s)
Here, Zn has changed into ZnSO4 (i.e., Zn2+ ions) by loss of electrons. Hence, Zn has been oxidised. CuSO4 (i.e., Cu2+) has changed into Cu by gain of electrons. Hence, CuSO4 has been reduced. Thus, the above reaction is a displacement reaction as well as a redox reaction.
(b) (i) Neutralisation reaction
(ii) Oxidation reaction.
Question 27.
When food materials containing fats and oils are left for a long time, what occurs? List two observable changes and provide three suggestions for preventing this phenomenon. (2020)
Answer:
Food materials containing fats and oils change their taste and smell due to a process called rancidity. Rancidity is a process in which air reacts with fats and oils which changes the smell and taste of food.
Methods of prevention:
Vacuum packing,
refrigeration of food materials,
placing of food materials, away from direct sunlight.
Question 28.
(i) Why is respiration considered as an exothermic reaction?
(ii) Write chemical name and the formula of the brown gas produced during thermal decomposition of lead nitrate.
(iii) Why do chips manufactures flush bags of chips with gas such as nitrogen? (Board Term I, 2015)
Answer:
(i) The glucose produced in our body during digestion combines with oxygen in the cells of our body and provides energy. The special name of this reaction is respiration. Thus, respiration is an exothermic process because energy is produced during this process.
C6H12O6(aq) + 6O2(g) → 6CO2(g) + 6H2O(l) + Energy
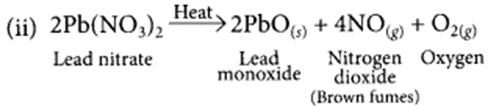
Brown gas evolved is nitrogen dioxide (NO2).
(iii) Chips manufacturers usually flush bags of chips with gas such as nitrogen because atmospheric oxygen can react with chips which may cause change in colour, change in taste. So, to cut the contact between air and the chips, nitrogen gas is used which do prevent its oxidation.
Short Answer Type Questions[l] [2 Marks] -Year 2015
29.“We need to balance a skeletal chemical equation.” Give reason to justify the statement.
Answer:
Skeletal chemical equation are unbalanced. We need to balance chemical equation because of law of conservation of mass. It states that ‘matter can neither be created nor be destroyed’. Therefore, chemical equation must be balanced in each and every chemical reaction.
30. Name the reducing agent in the following reaction:
3MnO2 + 4Al———— > 3Mn + 2Al2O3
State which is more reactive, Mn or A1 and why?
Answer. ‘Al’ is reducing agent.
‘AT is more reactive than Mn v ‘Al’ displaces Mn from its oxide.
Short Answer Type Questions[ll] [3 Marks] -Year 2015
31.A Name the type of chemical reaction represented by the following equation:
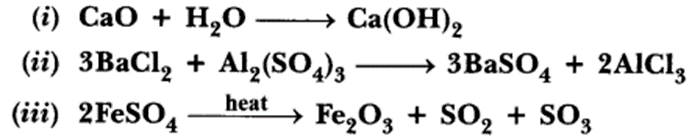
Answer.
(i) Combination reaction
(ii) Double displacement reaction (Precipitation reaction)
(iii) Decomposition reaction.
32. Write the chemical equation of the reaction in which the following changes have taken place with an example of each:
(i) Change in colour
(ii) Change in temperature
(iii) Formation of precipitate
Answer.
(i)Cu (s) + 2AgNO3 (aq)———–> Cu(NO3)2(aq) + 2Ag
The solution will become blue in colour and shiny silver metal will be deposited.
(ii) NaOH + HCl ———–> NaCl + H2O+ heat
The temperature will increase because heat will be evolved.
(iii) Pb(NO3)2 (aq) + 2KI (aq)———–> Pbl2 (s) + 2KNO3 (aq)
Yellow ppt
Yellow precipitate of Pbl2will be formed.
33.State the type of chemical reactions and chemical equations that take place in the following:
(i) Magnesium wire is burnt in air.
(ii) Electric current is passed through water.
(iii) Ammonia and hydrogen chloride gases ‘are mixed.
Answer.